
The port chambers without compromise
Advantages of PHS port systems: reduced risk of infection or occlusion, safe, economical
In the corners of port chambers of conventional standard ports, depositions of blood clots and drug residues can form. These often cause infection or occlusion (blockage) of the port system.
To minimize these risks, the PHS NuPort® systems have a significantly improved patented chamber geometry. The innovative elliptical inner chamber and the tangential outlet in the Nu-Flow® chambers significantly reduce the risk of these depositions due to their improved flushing characteristics.
Only 70% of the flushing volume is required for residue-free purging of the port chamber compared to a conventional standard port.

the most effective solution for long-term vascular access made from titanium
- suitable for high pressure injection (contrast-CT compatible)
- greatly reduced risk of occlusion and infection
- significantly improved flushing efficiency
- minimal maintenance
- facilitates blunt preparation of the port pocket
- MRI compatible up to 3-Tesla
- multiple catheter options

The economical solution for long-term vascular access made from titanium
- reduced risk of occlusion and infection
- improved flushing efficiency
- low maintenance
- MRI compatible up to 3-Tesla
- multiple catheter options

an effective solution for long-term vascular access made from polysulfone
- suitable for high pressure injection (contrast-CT compatible)
- reduced risk of occlusion and infection
- improved flushing efficiency
- low maintenance
- facilitates blunt preparation of the port pocket
- MRI compatible up to 3-Tesla
- multiple catheter options
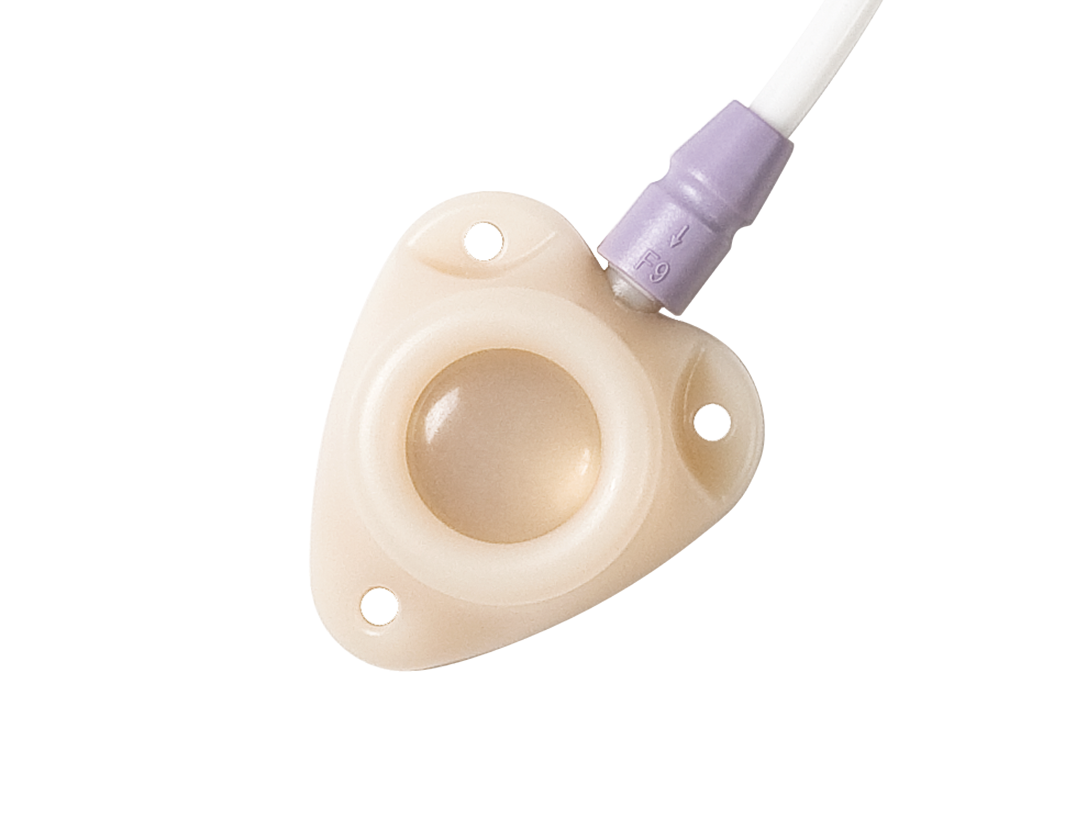
The economical solution for long-term vascular access made from polysulfone
- reduced risk of occlusion and infection
- improved flushing efficiency
- low maintenance
- facilitates blunt preparation of the port pocket
- MRI compatible up to 3-Tesla
- multiple catheter options
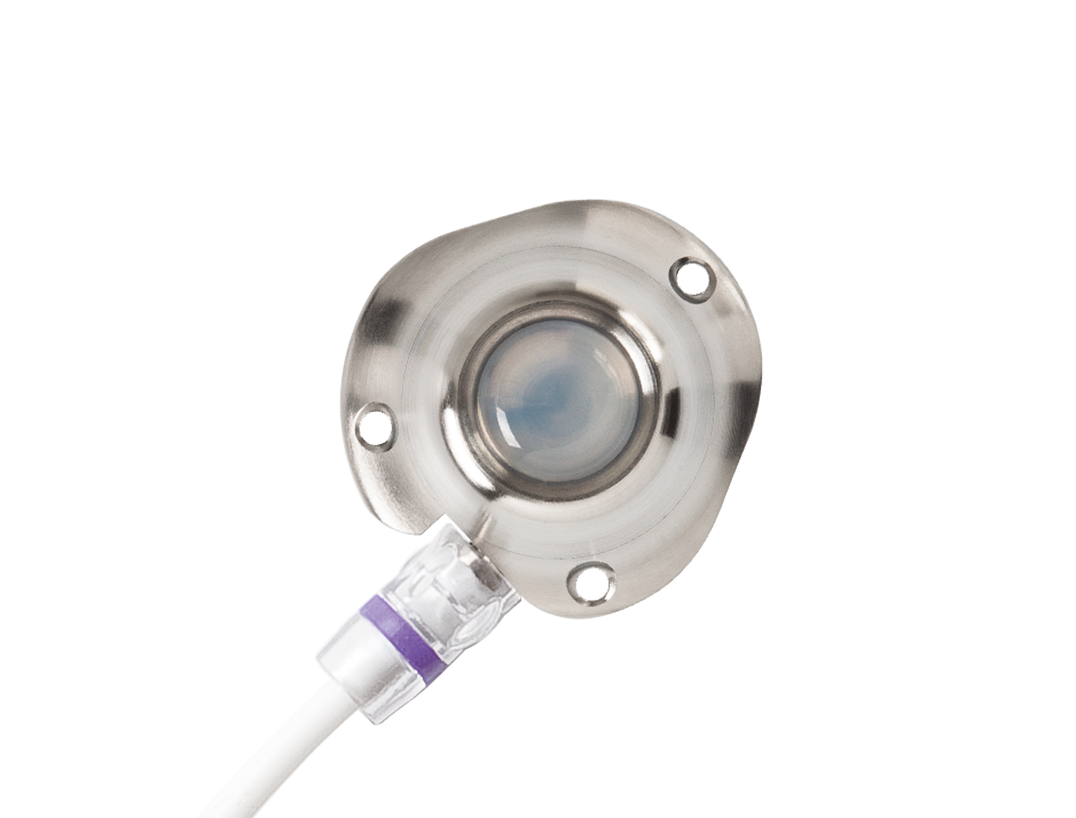
The advantages of the NuPort®-LP-CT reduced to a minimum in size.
- suitable for high pressure injection (contrast-CT compatible))
- optimized for cachectic and small patients
- greatly reduced risk of occlusion and infection
- significantly improved flushing efficiency
- minimal maintenance
- facilitates blunt preparation of the port pocket
- MRI compatible up to 3-Tesla
- multiple catheter options

- Optimized for PHS Medical GmbH port systems
- One-handed advancement of the guide wire possible using the thumb
- Introducer can be rotated 360° on dispenser
- Firm hold of the introducer needle on the guidewire straightener
- Smooth transition from dilator to sheath
- Tunneler with atraumatic tip
- Syringe with excentric luer




